

 Metal Injection Molding (MIM) product development from wide range of metallic materials; Stainless Steel 316L, Cobalt-Chrome and Titanium Alloys Ti6Al4V
Metal Injection Molding (MIM) product development from wide range of metallic materials; Stainless Steel 316L, Cobalt-Chrome and Titanium Alloys Ti6Al4V
Metal Injection Molding (MIM) lab offers a service of product development that has guarantee of a sintered parts that able to meet the minimum standard of Metal Powder Industries Federation Standard (MPIF) 35 in terms of physical and mechanical properties of the sintered MIM part and supported by a good results of microstructure behavior.
We have the expertise to produce these MIM technology based on more than 20 years of experience in R&D of Metal Injection Molding and countless testing and trial to produce the optimum results.
With our modern machines and production facilities, we can develop product in wide range of capacity; from lab scale to medium production scale to meet customer’s demand.
Standard procedure Metal Powder Industries Federation Standard (MPIF) 35
 Metal Injection Molding (MIM) Feedstock from wide range of metallic materials; Stainless Steel 316L, Cobalt-Chrome and Titanium Alloys Ti6Al4V
Metal Injection Molding (MIM) Feedstock from wide range of metallic materials; Stainless Steel 316L, Cobalt-Chrome and Titanium Alloys Ti6Al4V
Metal Injection Molding (MIM) lab offers a service of supplying MIM feedstock that has guarantee of a sintered parts that able to meet the minimum standard of Metal Powder Industries Federation Standard (MPIF) 35 in terms of physical and mechanical properties of the sintered MIM part and supported by a good results of microstructure behavior.
We have the expertise to produce these MIM feedstock based on more than 20 years of experience in R&D of Metal Injection Molding and countless testing and trial to produce the optimum results.
With our modern machines and production facilities, we can produce feedstock in wide range of capacity; from lab scale to medium production scale to meet customer’s demand.
Standard used:
* Standard procedure of mixing process
* Standard procedure of pelletizing process
Sintering and Heat Treatment Process in Iron, Steel, Nickel-Base, and Cobalt-Base Alloys and in Refractory and Reactive Metals
Metal Injection Molding (MIM) lab offers a service of running sintering and heat treatment process for a wide range of primary steels, ores, finished metals, and other inorganic materials.
We have the expertise to perform these processes at several conditions ranging from low to highest vacuum levels that can reach until 1.0 x 10-5 torr or under flowing of inert gas.
Heat rate can be ranging from the lowest of 0.5 °C/min and achieve the highest heating temperature of 1500 °C in our High Temperature Control Atmosphere Furnace (HTCAF).
Standard procedure of sintering process
Standard procedure of heat treatment process

* 3D Printing Technology
* Material Jetting or Polyjet
* Fused Deposition Modeling (FDM)
* Stereolithography (SLA)
* 3D printing has become a leading manufacturing technique in healthcare, engineering and medicine for a wide range of applications including dentistry, medical devices, anatomical models, engineered tissue models, aerospace, automotive and home appliances.

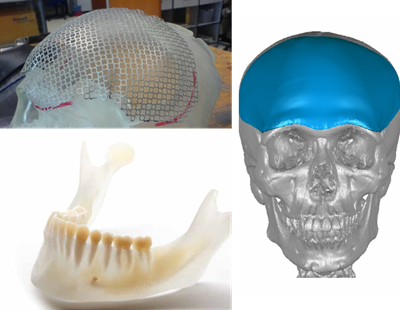 Biomodelling Services : -
Biomodelling Services : -
* Preformed Cranial Titanium Implants
* Anatomical Models for Surgical Planning
IC-Innovation Biomedical initiates and delivers innovative applications and solutions for Craniofacial Implant Services and Anatomical Models for Surgical Planning. With re-constructive surgery being a delicate art, medical professionals are now capable of meeting the challenge of treating patients without substantial destruction to human tissue. It also comes with the prospect of offering aesthetically pleasing results.
 Plasma spray is a thermal spray coating process used to produce a high-quality coating by a combination of high temperature, high energy heat source, a relatively inert spraying medium, usually argon, and high particle velocities. This technique can be used for ceramic coatings with desirable properties such as dense, resistant to wear, corrosion & heat, and to most acids, alkalis and solvents.
Plasma spray is a thermal spray coating process used to produce a high-quality coating by a combination of high temperature, high energy heat source, a relatively inert spraying medium, usually argon, and high particle velocities. This technique can be used for ceramic coatings with desirable properties such as dense, resistant to wear, corrosion & heat, and to most acids, alkalis and solvents.
Types of ceramic coatings: Aluminium oxide, Titanium oxide, Alumina-Titania, Zirconia base and Hydroxyapatite.
 This course of polymerization chemistry training and certification programme improve technical skill and productivity of participant in production, testing and troubleshooting in polymerization reactor. This course covers on material preparation and characterization in chemical and physical properties of polymer synthesized. Participants also experienced the online material check and troubleshooting of the reactor
This course of polymerization chemistry training and certification programme improve technical skill and productivity of participant in production, testing and troubleshooting in polymerization reactor. This course covers on material preparation and characterization in chemical and physical properties of polymer synthesized. Participants also experienced the online material check and troubleshooting of the reactor
TRAINING MODULE
* Introduction to the course
* Polymer chemistry
* Physical characterization of polymers
* Laboratory scale of polymerization
* Pilot Scale polymerization
* Characterization of polymers
OUTCOME
* Improve technical knowledge to carry out the polymerization
* Hands-on opportunity in polymerization and characterization
* Explore the art of science in preparing, interpreting and analysing polymers
* Exchange knowledge and ideas in polymerizations
 Freeze drying is the most suitable drying technique for substances that are non-resistance to high temperature in conventional drying. This technique is applied in pharmaceutical, cosmeceutical, natural product as well as nutritional food & beverage research and industry. It is also a well known technique to preserve the nutrition, composition and quality.
Freeze drying is the most suitable drying technique for substances that are non-resistance to high temperature in conventional drying. This technique is applied in pharmaceutical, cosmeceutical, natural product as well as nutritional food & beverage research and industry. It is also a well known technique to preserve the nutrition, composition and quality.
The concept of lyophilization in freeze drying / low temperature drying is about pre-freezing the sample and further eliminating the ice particle while maintaining most of the composition of the pre-frozen sample. The mechanism involved in this process is called sublimation (transition elements in solid form into vapour form without passing through an intermediate liquid phase).
Previously, we have been freeze drying various type of samples such as polymeric and ceramic materials as well as natural product ie. collagen, chitosan, bird’s nest, henna extract, blue pea extract, carrot, lime and others.
| 1) Polymer in solution form (before) and polymer in sponge form (after) | 2) Blue pea/bunga telang in extract solution form (before) and dried powder (after) | |||
| Before | After | Before | After | |

|

|

|

|
|

Our chemical reactor is a modular design laboratory reactor that function for optimization and simulation of various chemical reactions as well as for mixing and homogenizing processes in a laboratory scale. This system consist of stand system, laboratory stirring unit with power of high torque, anchor stirrer with flow boring, safety shutdown and reactor cover. Other device or apparatus such as dispersing unit, temperature sensors, flow breakers and other equipment can be installed on the free connections of the reactor cover. It is also suitable for vacuum operation and the seals that in contact with the product are made of solvent- respiratory temperature-resistant perfluoroelastomer (FFPM). Infinitely it can be adjustable on the speed. Besides, this reactor is also equipped by integrated torque trend display which is important for the measurement of viscosity changes. Through control actuated by microprocessor, the set speed is capable to be run in constant, even under load.

Cell proliferation is the increase in cell number as a result of cell growth and division. Cell proliferation study gives an informative result on whether a material influence on the cell growth and either cell numbers are increase or decrease with time.
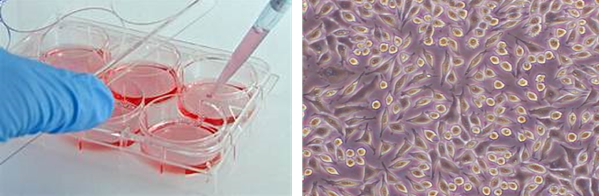
* In vitro toxicity testing is the scientific analysis of the effects of toxic chemical substances on mammalian cells.
* ISO 10993-5:2009; Biological evaluation of medical devices — Part 5: Tests for in vitro cytotoxicity.
* These methods specify the incubation of cultured cells in contact with a device and/or extracts of a device either directly or through diffusion. It is designed to determine the biological response of mammalian cells in vitro using appropriate biological parameters.

Observation / customization on plasma spray parameters and powder feedstock will be conducted in order to obtain optimum coating properties.

 Characterizations of plasma sprayed hydroxyapatite which involve phase content, crystallinity, surface morphology, microstructure and mechanical properties (SEM, EDX, XRD and adhesion).
Characterizations of plasma sprayed hydroxyapatite which involve phase content, crystallinity, surface morphology, microstructure and mechanical properties (SEM, EDX, XRD and adhesion).
ASTM F2024 - 10(2016) : Standard Practice for X-ray Diffraction Determination of Phase Content of Plasma-Sprayed Hydroxyapatite Coatings
This practice is for the determination, by the Reference Intensity Ratio External Standard Method, of the percent by weight of the crystalline phases, hydroxyapatite (HA), beta-(whitlockite) tricalcium phosphate (β-TCP), and calcium oxide (CaO) in coatings deposited upon metallic substrates by plasma-spraying hydroxyapatite
 Characterization for hydrophilic coating on the catheter in order to control all the factors that affect the coating integrity, which involves the composition of the coating and also observation of the quality of the surface being coated (SEM, EDX, XRD, FTIR, etc.)
Characterization for hydrophilic coating on the catheter in order to control all the factors that affect the coating integrity, which involves the composition of the coating and also observation of the quality of the surface being coated (SEM, EDX, XRD, FTIR, etc.)
 Calcium phosphate materials are used in many medical and dental applications such as coatings, fillers and so on. These materials are similar to bone in composition and having bioactive and osteoconductive properties. Calcium phosphate material will be synthesized using wet chemical technique. Characterizations of the synthesized powder will also be conducted using - X-ray diffractometer (XRD), Field Emission Scanning electron Microscope (FESEM), Energy Dispersive X-ray (EDX) and Fourier Transformation Infra-red (FTIR)
Calcium phosphate materials are used in many medical and dental applications such as coatings, fillers and so on. These materials are similar to bone in composition and having bioactive and osteoconductive properties. Calcium phosphate material will be synthesized using wet chemical technique. Characterizations of the synthesized powder will also be conducted using - X-ray diffractometer (XRD), Field Emission Scanning electron Microscope (FESEM), Energy Dispersive X-ray (EDX) and Fourier Transformation Infra-red (FTIR)
;ASTM F1609 - 08(2014): This specification covers the material requirements for calcium phosphate coatings for surgical implant applications. In particulate and monolithic form, the calcium phosphate materials system has been well-characterized regarding biological response and laboratory characterization. This specification includes hydroxylapatite coatings, tricalcium phosphate coatings, or combinations thereof, with or without intentional minor additions of other ceramic or metallic, and applied by methods including, but not limited to, the following: mechanical capture, plasma spray deposition, dipping/sintering, electrophoretic deposition, porcelainizing, and sputtering. Substrates may include smooth, porous, textured, and other implantable topographical forms. This specification excludes organic coatings that may contain calcium and phosphate ionic species. Materials shall be tested and the individual grades shall conform to chemical requirements such as elemental analysis fo r calcium and phosphates, and intentional additions, trace element analysis for hydroxylapatite and beta tricalcium phosphate; crystallographic characterization such as Fourier Transform infrared spectroscopy, and environmental stability; physical characterization such as coverage of substrate, thickness, porosity, color, surface topography, and density; and mechanical characterization such as tensile bond strength, shear strength, and fatigue strength. The test specimen fabrication and contact with calcium phosphate coatings are also detailed.